Overview
This article reviews the dermatologic manifestations of Mycobacterium avium-intracellulare (MAI; also referred to as M avium complex [MAC]) infection.
MAI is an opportunistic pathogen that usually causes disease in the weakened immune system. [1] Although MAI infection has become more prevalent since the AIDS epidemic, it remains a rare cause of skin disease. MAI has been found to cause cutaneous disease mostly in individuals with immunosuppression, though it has also been reported in immunocompetent patients. [1]
Primary cutaneous MAI infection generally occurs in hosts who are immunocompetent after traumatic introduction into the soft tissue. Local disease ensues, manifested primarily as nodules that may progress to ulcers. One case report described primary MAI infection resulting in a clinical picture resembling lupus vulgaris. [2]
Disseminated MAI (DMAI) infection most commonly occurs in individuals who are immunosuppressed, and hematogenous seeding of the skin may result in a wide variety of skin lesions, including panniculitis, folliculitis, acute disease with hemorrhagic pustules, nodules, and abscesses. Disease may present primarily in the skin with later dissemination. [3]
See the image below.
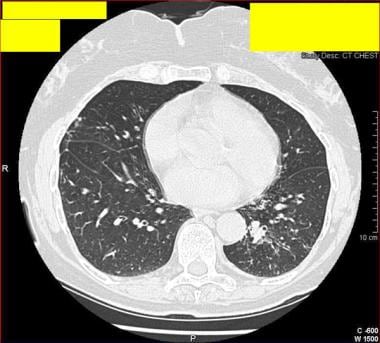 CT thorax scan of a 77-year-old woman who presented with chronic cough and sputum production, without a history of underlying preexisting lung disease. Sputum culture grew Mycobacterium avium complex. The diagnosis was Lady Windermere syndrome.
CT thorax scan of a 77-year-old woman who presented with chronic cough and sputum production, without a history of underlying preexisting lung disease. Sputum culture grew Mycobacterium avium complex. The diagnosis was Lady Windermere syndrome.
MAI is the most common cause of pediatric cervical adenitis. Skin involvement in this setting is not uncommon and includes sinus track and abscess formation. Cervical adenitis rarely involves deeper structures, but it may result in extensive superficial destruction with cosmetic deformity.
See also Mycobacterium avium Complex (MAC) (Mycobacterium avium-intracellulare [MAC]).
Epidemiology
In the United States, MAI (MAC) infection is considered a nonreportable infectious disease. US Centers for Disease Control and Prevention (CDC) surveillance data from Houston and Atlanta suggested an annual incidence of 1 case per 100,000 persons. The incidence of DMAI peaked in 1994 at 37,000 cases, coinciding with the height of the AIDS epidemic. This coincidence concords with the finding that DMAI infection often occurs in association with HIV disease. One case series revealed cutaneous involvement in six of 30 cases of DMAI infection. CDC data also suggested that the incidence of MAI infection may be decreasing as a result of highly active antiretroviral therapy and antimicrobial prophylaxis. [4, 5]
Primary cutaneous MAI infection is rare, with only 12 cases having been reported in the literature before 1997. This condition has been reported in all age groups; although most reported cases have been in individuals aged between 2 and 10 years, adult cases have also been described. A review of eight cases of cutaneous MAI infection occurring secondary to disseminated disease found a median patient age of 22.2 years (range, 2-28 y). A 2018 review of 30 cases of cutaneous MAI reported an average age of 44.2 years (range, 4-82 y); of these, eight patients were younger than 18 years. [1]
The degree to which skin involvement occurs as a sequela of cervical adenitis has not been well studied; however, the literature has suggested that the risk of skin involvement increases with advanced disease. Cervical adenitis most commonly affects children. One chart review series of 47 children with cervical adenitis revealed a median age of 3 years at the time of MAI infection diagnosis. [6]
Clinical Evaluation
It is important to consider cutaneous MAI (MAC) infection in patients with antibiotic-resistant cellulitis, nonhealing nodules, and ulcers, as well as in patients who are immunosuppressed with evidence of DMAI infection.
Data on skin and soft-tissue infection caused by nontuberculous mycobacteria in Taiwan from the period 1997-2008 suggested that MAI is an important pathogen. [7]
Any history of the introduction of a foreign object (eg, needle, splinter) should be sought if MAI infection is suspected. Fever and other constitutional symptoms are generally absent. Skin abscess and sinus tracks may be present. Sporotrichoid spread of cutaneous MAI infection can occur. [8]
Patients with MAI cervical adenitis may present with an indolent course of a unilaterally expanding mass in the neck, such as enlarged submandibular or submaxillary nodes.
Cutaneous disease
Cutaneous MAI infection should be suspected when an individual who is immunocompromised presents with obscure skin lesions. Additional symptoms may suggest disseminated disease, including fatigue, fever, diarrhea, weight loss, back or bone pain, shortness of breath, and cough. Other clinical manifestations include involvement of organs such as the lungs, lymphatic system, [9] and musculoskeletal system. [1]
A 2024 case study illustrated the diagnostic complexity of cutaneous MAI infection, which was initially misdiagnosed as sarcoidosis in an immunosuppressed adolescent with persistent skin lesions. [10] Early biopsies, which showed intact reticulin fibers but no organisms, suggested sarcoidosis; however, subsequent analysis revealed acid-fast organisms and confirmed MAI infection via polymerase chain reaction (PCR) assay. This study highlighted the importance of repeated biopsies and molecular diagnostics in distinguishing MAI from similar granulomatous conditions, given that traditional markers like reticulin fiber integrity may be unreliable.
Nutritional status impacts susceptibility to MAI infection. A case report describing a patient with anorexia nervosa who also developed MAI and leukocytoclastic vasculitis was notable but not altogether surprising. [11]
Cutaneous MAI infection has also been reported to mimic histoid leprosy in patients with HIV infection or AIDS. [12, 13] Subcutaneous and muscular abscesses due to MAI can occur as a manifestation of immune restoration in patients with AIDS. [14] Other reports have included a case mimicking sarcoid [15] and a case reporting a cutaneous pseudotumor related to MAI. [16]
Primary cutaneous MAI infection is most often the result of traumatic inoculation, though a specific history of trauma is generally absent. Patients may describe the development of a single nodule or multiple nodules progressing over time into ulcerative lesions with or without serosanguineous exudates. Painful subcutaneous nodules and ulcers that occur on the extremities and the trunk may be noted. Lesions may be present in multiple stages of development, and nodules have been described as both flesh-colored and purple-red.
MAI has also been noted to be associated with several diverse pathologies. Teraki et al noted that MAI can be associated with Sweet syndrome. [17] Henoch-Schönlein purpura associated with pulmonary MAI infection has also been noted. [18]
MAI can be associated with osteomyelitis as well. Primary nontraumatic MAI osteomyelitis of the distal phalanx has occurred. [19] Multifocal osteomyelitis caused by nontuberculous mycobacteria in patients with a genetic defect of the interferon gamma receptor has also been noted.
Murdoch and McDonald reported MAI cellulitis occurring with septic arthritis after joint injection. [20]
After initiation of treatment for MAI infection involving osteomyelitis, DRESS (Drug Reaction with Eosinophilia and Systemic Symptoms) syndrome (also referred to as DIHS [Drug-Induced Hypersensitivity Syndrome]) has been reported to occur. [21]
MAI olecranon bursitis resolving without surgical or antimicrobial intervention has been observed. [22]
A 2018 review of 30 cases found that patients had associated medical histories of organ transplantation, hemodialysis, underlying malignancies, and pulmonary tuberculosis. [1]
A 2021 case report described MAI infection associated with concurrent small-bowel obstruction, which indicated the possibility of MAI infection in cases of abdominal pain occurring surgery for small-bowel obstruction. [23] Another report from 2020 found extranodal MAI infection, likely from hot tubs, in two immunocompetent children, with primary involvement in the pulmonary and urinary systems. [24]
A 2020 review found MAI to be an emerging pathogen in dog breeders with hereditary immunodeficiencies, particularly those who breed dogs with a predisposition for MAI infection (eg, miniature schnauzers and basset hounds). The review supported the zoonotic potential of canine MAI infections to be transmitted to immunocompromised human patients. [25]
Workup Considerations
Primary cutaneous MAI (MAC) infection is rare, but it does not necessarily mandate a workup for immunosuppression.
Biopsy should be performed for suspected dermatologic manifestations of MAI infection. Tissue samples of cutaneous lesions may be obtained for histopathologic evidence of mycobacterial infection, and staining with Ziehl-Neelsen stain may reveal acid-fast bacilli (AFB). [1]
Faruqi et al noted that a positive antineutrophil cytoplasmic antibody (ANCA) test result can coexist with MAI, complicating diagnosis. [26]
Cultures of cutaneous lesions should be performed, and PCR assays of tissue may yield organism identification in 24 hours.
A 2012 report described a 66-year-old women who developed sudden redness and swelling of her left thumb. [27] Histology showed well-formed granulomas along with a mixed inflammation. The infiltrate was characterized by a predominance of neutrophils, and stains showed AFB. Culture results were negative. The pathogenic mycobacterium was identified through broad-range PCR amplification followed by suspension array identification.
18F-Fluorodeoxyglucose (FDG) positron emission tomography (PET) can be used to distinguish DMAI infection from paraneoplastic syndromes or malignant metastasis. [28] Molecular assays might be useful in some cases. [5]
A 2020 case report described using magnetic resonance imaging (MRI) for evaluation of a horseshoe abscess in order to diagnose and determine the extent of a hand infection. [29] MRI revealed rice bodies secondary to MAI infection. MRI may provide initial data for consideration of infection with mycobacterial organisms.
Histologic Features
A spectrum of histologic findings is present in skin infection with MAI (MAC). Tissue pathology varies with the stage of the disease and the host's immune status.
Granulomas are often present in tissue infected with MAI. Tuberculid, palisading, and sarcoidlike granulomas can be found in the context of a diffuse infiltrate of foamy histiocytic cells. Other nonspecific findings include panniculitis, chronic inflammation, cutaneous abscesses, necrotizing folliculitis, and suppurative granulomas.
Spindle cell pseudotumors
Liou et al noted that spindle cell pseudotumors due to mycobacterial infection may occur in immunocompromised hosts. [30] The investigators reported the case of a 37-year-old man with AIDS in whom a spindle cell pseudotumor manifested as a firm nodule on the right arm. Histologically, the tumor was composed of spindle cells interspersed with histiocytes and inflammatory cells. Ziehl-Neelsen stain revealed many AFB identified as MAI. Culture and sequencing of the PCR product of the mycobacterial 65-kd heat-shock protein gene helped establish the diagnosis. Immunohistochemically, the spindle cells stained with CD68, a marker found on active tissue macrophages.
Similarly, Shiomi et al reported the case of a 58-year-old Japanese woman with systemic lupus erythematosus (SLE) and interstitial pneumonia for 17 years who was diagnosed with mycobacterial spindle cell pseudotumor. [31] The patient had a nodule in the deep dermis with extension into the subcutaneous fat tissue. Under histologic examination, the nodule was composed of spindle cells focally showing a vaguely storiform pattern, with focal foam cells, epithelioid histiocytes, and multinucleated giant cells. Ziehl-Neelsen staining showed numerous AFB within the spindle cells and epithelioid histiocytes, which were determined to be MAI after culture and PCR testing.
Treatment and Monitoring Considerations
There is some evidence that bacille Calmette-Guérin (BCG) vaccination can induce cross-immunity to MAI (MAC). [32]
Patients hospitalized with MAI disease require inpatient care. However, primary cutaneous MAI infection and cervical adenitis may be treated in an outpatient setting if patients are medically compliant.
A standard chemotherapy regimen has not been established for MAI infection, because of significant resistance to antimycobacterial drugs. MAI is usually resistant to single-drug therapy and must be treated with multiple antitubercular medications. Sparfloxacin has good antimicrobial activity against several AFB and is expected to be an effective drug for treating mycobacteriosis.
MAI infection can be related to infliximab use. [33] MAI infections have been associated with therapy with brentuximab vedotin. [34]
In cases of cervical adenitis and primary cutaneous MAI infection, follow-up care is required to assess for toxicity due to therapeutic agents and to monitor patients for possible disease recurrence or surgical complications.
Also see Guidelines for the prevention and treatment of opportunistic infections in HIV-exposed and HIV-infected children: recommendations from the National Institutes of Health, Centers for Disease Control and Prevention, the HIV Medicine Association of the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the American Academy of Pediatrics. [4]
Management of Cervical Adenitis
All cases of cervical adenitis must be referred to a surgeon.
The mainstay of treatment for cervical adenitis due to MAI (MAC) infection is surgical excision, with chemotherapy as primary treatment mainly reserved for cases with surgical contraindications. [35] Vuppalapati et al reported treating MAI infection involving the skin and soft tissues of the hand with radical debridement and reconstruction in addition to multidrug chemotherapy. [36]
The relative infrequency of this condition has precluded clinical trials; therefore, treatment schedules are derived from anecdotal evidence.
Macrolides should be used in combination with rifamycin or ethambutol. Combination therapy with clarithromycin, ethambutol, and rifamycin has also been used.
Management of Primary Cutaneous MAI Infection
In a case report involving a 9-year-old patient with MAI (MAC) infection, treatment with a combination of surgery and cycloserine 500 mg/day, isoniazid 200 mg/day, and clarithromycin 150 mg/day following susceptibility testing resulted in complete clearance after 2 months of medical therapy followed by surgical excision. [37]
For adult patients, streptomycin 1 g/day intramuscularly (IM) for 3 months, isoniazid 300 mg/day, and rifampicin 600 mg/day for 9 months have been used as treatment. Some cases also require local excision and skin grafting. [1] A case study from 2019 reported using vacuum-sealing drainage at the surgical incision site to drain pathogens and secretions from the infected location. [38]
Outcomes
Primary cutaneous involvement of MAI (MAC) usually does not result in systemic disease and most often occurs in otherwise healthy individuals; therefore, the prognosis is favorable, because treatment is generally curative. One review of 30 cases found that treatment duration can range from 2 to 16 months. [1]
The relation between cutaneous lesions and mortality in DMAI infection is unknown, and controversy remains regarding the impact of DMAI infection on life expectancy in patients with AIDS. Nonetheless, untreated DMAI infection in this population (patients with AIDS) is clearly associated with increased mortality.
Primary cutaneous MAI infection and atypical Mycobacterium-induced cervical adenitis are responsive to combined surgical and medical treatment. However, cervical adenitis may cause local destruction of superficial structures and may result in significant cosmetic and functional impairment without treatment. Dissemination beyond the primary site is rare. Nonetheless, the proximity to the trachea and the oropharynx may result in airway compromise and should be considered in the evaluation of patients with cervical adenitis.
-
CT thorax scan of a 77-year-old woman who presented with chronic cough and sputum production, without a history of underlying preexisting lung disease. Sputum culture grew Mycobacterium avium complex. The diagnosis was Lady Windermere syndrome.

