Practice Essentials
Basal cell carcinoma (BCC) is a nonmelanocytic skin cancer (ie, an epithelial tumor) that arises from basal cells (ie, small, round cells found in the lower layer of the epidermis). [1, 2] The prognosis for patients with BCC is excellent, but if the disease is allowed to progress, it can cause significant morbidity. [3]
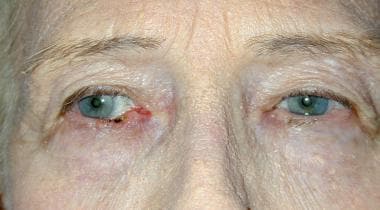
The image below depicts BCC of the right lower lid.
Signs and symptoms
Approximately 85% of BCCs occur on the face, head (scalp included), and neck; others appear on the trunk or extremities; rarely, they may occur on the hands. [3, 4] Other characteristic features of BCC tumors include the following:
-
Waxy papules with central depression
-
Pearly appearance
-
Erosion or ulceration: Often central and pigmented
-
Bleeding: Especially when traumatized
-
Oozing or crusted areas: In large BCCs
-
Rolled (raised) border
-
Translucency
-
Telangiectases over the surface
-
Slow growing: 0.5 cm in 1-2 years
-
Black-blue or brown areas
Periocular tumors most commonly involve the following:
-
Lower eyelid: 48.9-72.1%
-
Medial canthus: 25-30%
-
Upper eyelid: 15%
-
Lateral canthus: 5%
Clinicopathologic types of BCC, each of which has a distinct biologic behavior, include the following:
-
Nodular: Cystic, pigmented, keratotic; the most common type of BCC; usually presents as a round, pearly, flesh-colored papule with telangiectases
-
Infiltrative: Tumor infiltrates the dermis in thin strands between collagen fibers, making tumor margins less clinically apparent
-
Micronodular: Not prone to ulceration; may appear yellow-white when stretched, is firm to the touch, and may have a seemingly well-defined border
-
Morpheaform: Appears as a white or yellow, waxy, sclerotic plaque that rarely ulcerates; is flat or slightly depressed, fibrotic, and firm
-
Superficial: Seen mostly on the upper trunk or shoulders; appears clinically as an erythematous, well-circumscribed patch or plaque, often with a whitish scale
See Presentation for more detail.
Diagnosis
Given that BCC rarely metastasizes, laboratory and imaging studies are not commonly clinically indicated in patients presenting with localized lesions. Imaging studies may be necessary when involvement of deeper structures is clinically suspected (eg, CT for suspected bone involvement, MRI for soft-tissue or perineural ay be used for assessment of .
Biopsy
Types of skin biopsy that may be used to confirm the diagnosis and determine the histologic subtype of BCC include the following:
-
Shave biopsy: Most often, the only biopsy that is required
-
Punch biopsy: May be indicated in the case of a pigmented lesion if there is difficulty distinguishing between pigmented BCC and melanoma; ensures that the depth of the lesion can be determined if it proves to be a malignant melanoma
Histology
Histologically, BCC is divided into the following 2 categories:
-
Undifferentiated: When there is little or no differentiation, the carcinoma is referred to as solid BCC; this form includes pigmented BCC, superficial BCC, sclerosing BCC, and infiltrative BCC (a histologic subtype)
-
Differentiated: Differentiated BCC often has slight differentiation toward hair (keratotic BCC), sebaceous glands (BCC with sebaceous differentiation), and tubular glands (adenoid BCC); noduloulcerative (nodular) BCC is usually differentiated
See Workup for more detail.
Management
Surgery
In nearly all cases of BCC, surgery is the recommended treatment modality. [5, 6] Techniques used include the following [7] :
-
Electrodesiccation and curettage
-
Excisional surgery
-
Mohs micrographically controlled surgery
-
Cryosurgery
Radiation therapy
BCCs are usually radiosensitive; radiation therapy (RT) can be used in patients with advanced and extended lesions, as well as in those for whom surgery is not suitable. Postoperative radiation can also be a useful adjunct when patients have aggressive tumors that were treated surgically or when surgery has failed to clear the margins of the tumor. [8]
Photodynamic therapy
Photodynamic therapy (PDT) as an adjunct is a reasonable choice in the following cases:
-
Tumor recurrence with tissue atrophy and scar formation
-
Elderly patients or patients with medical conditions preventing extensive oculoplastic reconstructive surgery
-
Tumor with poorly defined borders based on clinical examination
-
Tumor requiring difficult or extensive oculoplastic surgery
Pharmacologic therapy
Topical agents used in the treatment of superficial BCC include the following [7] :
-
Topical 5-fluorouracil 5%: May be used to treat small, superficial BCCs in low-risk areas
-
Imiquimod: May be used for the treatment of nonfacial superficial BCC
-
Tazarotene: Can also be used to treat small, low-risk BCCs
Oral agents approved by the US Food and Drug Administration (FDA) for advanced forms of BCC include the following hedgehog pathway inhibitors (HHIs):
-
Vismodegib (Erivedge)
-
Sonidegib (Odomzo)
The checkpoint inhibitor cemiplimab (Libtayo) is approved for patients with locally advanced BCC and has been granted accelerated approval for use in patients with metastatic BCC previously treated with an HHI or for whom an HHI is not appropriate.
See Treatment and Medication for more detail.
Background
Basal cell carcinoma (BCC) is the most common skin cancer in humans, yet it accounts for less than 0.1% of patient deaths from cancer. Basal cell skin cancer tumors typically appear on sun-exposed skin, are slow growing, and rarely metastasize (0.028-0.55%). BCC usually appears as a flat, firm, pale area that is small, raised, pink or red, translucent, shiny, and waxy, and the area may bleed following minor injury. Tumor size can vary from a few millimeters to several centimeters in diameter. (See Presentation.)
See the clinical images below of basal cell carcinoma.
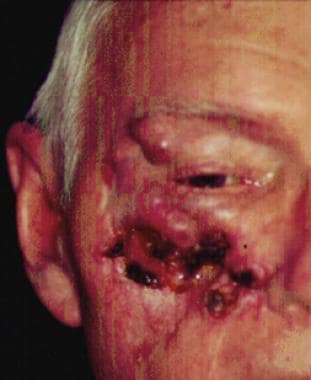 A 68-year-old patient presenting with an advanced basal cell carcinoma (BCC) of the right periorbital region, frontal view. Courtesy of M Abraham Kuriakose, DDS, MD.
A 68-year-old patient presenting with an advanced basal cell carcinoma (BCC) of the right periorbital region, frontal view. Courtesy of M Abraham Kuriakose, DDS, MD.
BCC is a nonmelanocytic skin cancer (ie, an epithelial tumor) that arises from basal cells, which are small round cells found in the lower layer of the epidermis. Basal cells invade the dermis but seldom invade other parts of the body. Most of the DNA alterations involved in BCC result from damage caused by exposure to sunlight, but inheritance may be a factor in some cases. (See Pathophysiology.)
Body distribution of BCCs is as follows:
-
Head and neck (most frequently on the face [9] ; most common location is the nose, specifically the nasal tip and alae) - 85%
-
Trunk and extremities [3] -15%
The anatomic distribution of BCCs correlates with embryonic fusion planes. After adjusting for surface area, BCC occurrence is more than 4 times more likely to develop on embryonic fusion planes than on other regions of the midface, a finding that supports the possibility of an embryologic role for BCC pathogenesis. [12]
BCC can develop on unexposed areas. In some patients, contributing factors are exposure to or contact with arsenic, [13] tar, coal, paraffin, [14] certain types of industrial oil, and radiation. BCC can also be associated with scars (eg, burn complications), [15] xeroderma pigmentosum, [16] , previous trauma. [17] vaccinations, or even tattoos. (See Etiology.)
A skin biopsy (most often a shave biopsy is sufficient) may be necessary to confirm the diagnosis and is often required to determine the histologic subtype of BCC. A punch biopsy may be used to obtain a thick specimen, especially when the clinical suspicion of a BCC is still present after shave biopsy results are negative. (See Workup.)
Neglected tumors can continue to grow and lead to significant local destruction and even disfigurement. Surgery, in almost all cases, is the recommended treatment, [5, 7] with treatments varying on the basis of cancer size, depth, and location. (See Treatment.)
Superficial BCCS have been successfully treated with imiquimod 5% cream, [18, 19] and topical 5-fluorouracil 5% cream may be used to treat small, superficial BCCs. [20] Several studies have shown success in treating small nodular BCCs with imiquimod 5% cream, although this is an off-label indication and patients should be informed of this fact. (See Medication.)
Pathophysiology
Although the exact etiology of BCC is unknown, a well-established relationship exists between BCC and the pilosebaceous unit, as tumors are most often discovered on hair-bearing areas.
Many believe that BCCs arise from pluripotential cells in the basal layer of the epidermis or follicular structures. These cells form continuously during life and can form hair, sebaceous glands, and apocrine glands. Tumors usually arise from the epidermis and occasionally arise from the outer root sheath of a hair follicle, specifically from hair follicle stem cells residing just below the sebaceous gland duct in an area called the bulge.
Signaling pathways
The patched/hedgehog intracellular signaling pathway plays a role in both sporadic BCCs and nevoid BCC syndrome (Gorlin syndrome). This pathway influences differentiation of various tissues during fetal development. After embryogenesis, it continues to function in regulation of cell growth and differentiation. Loss of inhibition of this pathway is associated with human malignancy, including BCC.
The hedgehog gene encodes an extracellular protein that binds to a cell membrane receptor complex to start a cascade of cellular events leading to cell proliferation. Of the three known human homologues, sonic hedgehog (SHH) protein is the most relevant to BCC. Patched (PTCH) is a protein that is the ligand-binding component of the hedgehog receptor complex in the cell membrane. The other protein member of the receptor complex, smoothened (SMO), is responsible for transducing hedgehog signaling to downstream genes. [21, 22]
When SHH is present, it binds to PTCH, which then releases and activates SMO. SMO signaling is transduced to the nucleus via Gli. When SHH is absent, PTCH binds to and inhibits SMO. Mutated forms of PTCH are unable to bind to SMO, simulating the presence of SHH. The unbound SMO and downstream Gli are constitutively activated, thereby allowing hedgehog signaling to proceed unimpeded. The same pathway may also be activated via mutations in the SMO gene, which also allows unregulated signaling of tumor growth.
How these defects cause tumorigenesis is not fully understood, but most BCCs have abnormalities in either PTCH or SMO genes. Some authors even consider defects in the hedgehog pathway to be requirements for BCC development.
BCC most commonly develops on sun-exposed areas. Zhang et al reported that ultraviolet (UV)-specific nucleotide changes in PTCH, as well as the tumor suppressor gene TP53, are implicated in the development of early-onset BCC. [23]
UV-induced mutations in the TP53 tumor suppressor gene, which resides on band 17p13.1, have been found in some cases of BCC. [24] A germline single-nucleotide polymorphism (SNP) in the TP53 gene, rs78378222, has also been associated with susceptibility to BCC. [24] In addition, frameshift mutations of the BAX gene (BCL2-associated X protein) have been found in sporadic cases of BCC. A reduction of bcl-2 proteins is observed in the aggressive, infiltrative type of BCC.
Radiation and immunologic origins
Radiation has proven to be tumorigenic by two mechanisms. The first entails the initiations of prolonged cellular proliferation, thereby increasing the likelihood of transcription errors that can lead to cellular transformation. The second mechanism is direct damage of DNA replication, leading to cellular mutation that may activate proto-oncogenes or deactivate tumor suppressor genes.
Immunologically, the mechanism by which prolonged ultraviolet radiation exposure leads to the development of BCC includes suppression of the cutaneous immune system and immunologic unresponsiveness to cutaneous tumors. This local effect includes a decrease in Langerhans cells, dendritic epidermal T cells, and Thy1+ cells. Furthermore, systemic proliferation of suppressor T cells and the release of immunosuppressive factors (eg, tumor necrosis factor–alpha [TNF-alpha], interleukin-1 [IL-1], prostaglandin [PG], IL-10) are believed to be involved in the development of BCC.
DNA mismatch repair proteins
DNA mismatch repair (MMR) proteins are a group of proteins that physiologically stimulate G2 cell cycle checkpoint arrest and apoptosis. Failure of MMR proteins to detect induced DNA damage results in the survival of mutating cells. MMR protein levels have been found to be higher in nonmelanoma skin cancers than in normal skin, and there is also some evidence of MMR dysregulation. [25]
Etiology
The exact cause of BCC is unknown, but environmental and genetic factors are believed to predispose patients to BCC.
Radiation exposure
Sunlight, particularly long-term exposure, is the most frequent association with development of BCC; risk correlates with the amount and nature of accumulated exposure, especially during childhood. Patient geographic location affects the risk of developing skin cancer. A latency period of 20-50 years is typical between the time of ultraviolet (UV) damage and BCC clinical onset.
The prevalence of BCC increases in areas of higher altitude and in areas of lower latitude. The incidence of BCC is rising, potentially because of atmospheric changes and the increased popularity of sunbathing.
Radiation exposure that contributes to BCC development may also include tanning booths and UV light therapy. Both short-wavelength UVB radiation (290-320 nm, sunburn rays) and longer wavelength UVA radiation (320-400 nm, tanning rays) contribute to the formation of BCC. UVB is believed to play a greater role in the development of BCC than UVA, however, and is the primary agent responsible for most skin cancer. [26]
UVB and UVC can modify unsaturated chemical bonds of nucleic acids, which may lead to mutations. UVC does not penetrate the atmospheric ozone layer. The UVA spectrum is absorbed by melanin and, through free-radical transfer, affects cellular deoxyribonucleic acid (DNA). Mutations caused by UV radiation typically include cytosine (C) to thymine (T), or CC to TT, translocation. This process can cause activation of oncogenes or inactivation of tumor suppressor genes, leading to tumor initiation and progression. [27]
The skin can repair superficial damage, but the underlying cumulative damage remains, including DNA damage. The damage worsens with each successive sun exposure, causing a lifetime progression. [28]
In a 2012 systematic review and meta-analysis of 12 studies with 9328 cases of nonmelanoma skin cancer, Wehner et al found that indoor tanning was associated with a significantly increased risk of both basal and squamous cell skin cancer. The risk was highest among users of indoor tanning before age 25. The authors estimate that the population-attributable risk fraction in the United States is 8.2% for squamous cell carcinoma and 3.7% for BCC, corresponding to more than 170,000 cases of nonmelanoma skin cancer annually caused by indoor tanning. [29] In another 2012 study of 376 patients with BCC and 390 control patients with minor benign skin conditions, indoor tanning was strongly associated with early-onset BCC, particularly in women. [30]
X-ray and Grenz-ray exposure are also associated with BCC formation.
Gene mutations
Studies have demonstrated a high incidence of TP53 gene mutations in BCC. Researchers speculate that ultraviolet sunlight may play an important role in the genesis of this mutation; yet, genetic involvement has been demonstrated on chromosome 9 only in patients with familial basal cell nevus syndrome (Gorlin syndrome). Such mutation involves the patched (PTCH) gene, a tumor suppressor gene.
Inappropriate activation of the hedgehog signaling pathway is found in both sporadic and familial cases of BCC. This results in loss-of-function mutations in tumor-suppressor protein patched homologue 1 (PTCH1) and gain-of-function mutations in sonic hedgehog (SHH), smoothened (SMO), and Gli.
Arsenic ingestion
Arsenic — particularly in the form of inorganic arsenic compounds — has been identified as carcinogenic in humans. High levels of arsenic occur naturally in drinking water in various parts of the world, including the western United States. [31] Arsenical skin lesions, which are the hallmark of chronic arsenic poisoning, are considered an indication of elevated skin cancer risk. [13]
Immunosuppression
A modest increase in the lifetime risk of BCC has been noted in chronically immunosuppressed patients, such as recipients of organ or stem cell transplants and people with AIDS. [32]
Organ transplant patients must be instructed to limit sun exposure and be alerted that skin cancer is a serious problem for them. In fact, immunosuppression and sun damage may cooperate to cause skin cancer. The skin cancer incidence is 10-fold higher in transplant recipients than in the general population; up to 65-75% of patients with long-term immunosuppression develop skin cancer. Transplant recipients who have who developed a posttransplant skin cancer are at elevated risk of developing a subsequent skin cancer within 2 years, with approximately 1 in 20 developing 10 or more skin cancers. [33]
Previous nonmelanoma skin cancer
Persons who have been diagnosed with one nonmelanoma skin cancer are at increased risk of developing tumors in the future. The risk of developing new nonmelanoma skin cancers is reported to be 35% at 3 years and 50% at 5 years after an initial skin cancer diagnosis. [34]
Skin type
Albinism has been implicated in BCC. The Fitzpatrick skin-type scale, which ranges from very fair (skin type I) to very dark (skin type VI), categorizes cutaneous sensitivity to ultraviolet radiation. It is based on the individual's tendency to burn and tan and is a good predictor of relative risk among Whites.
Alcohol consumption
A study among adults in the United States reports a strong association between excessive alcohol drinking and higher incidence of sunburn, suggesting a linkage between alcohol consumption and skin cancer. [35]
Hydrochlorothiazide use
The widely used diuretic hydrochlorothiazide (HCTZ) is a potent photosensitizer. Slightly increased risk for BCC (and dramatically increased risk for cutaneous squamous cell carcinoma [SCC]) was documented in a case-control study that cross-referenced 71,533 cases of BCC and 8629 cases of SCC from the Danish Cancer Registry with data on cumulative HCTZ exposure in these cases with that country’s National Prescription Registry. No association was found between nonmelanoma skin cancer and other antihypertensive drugs. [36]
The study results supported a dose-response relationship between HCTZ use and BCC, as follows [36] :
-
High cumulative use of HCTZ (≥50,000 mg) increased the odds ratio (OR) of BCC by 1.29.
-
Patients with the highest HCTZ exposure (> 200,000 mg) had an OR of 1.54 for BCC.
The association of BCC with HCTZ use was strongest in younger individuals (age 50 years and younger). In these patients, the OR was 1.91. Overall, the proportion of BCCs attributable to HCTZ use was 0.6%. HCTZ showed the strongest association with skin cancers on heavily sun-exposed sites such as the lower limbs (versus the trunk). [36]
Hereditary disorders
The following inherited conditions are associated with increased risk of BCC:
-
Xeroderma pigmentosum - This autosomal recessive disease results in the inability to repair ultraviolet-induced DNA damage. Pigmentary changes are seen early in life, followed by the development of BCC, squamous cell carcinoma, and malignant melanoma. Other features include corneal opacities, eventual blindness, and neurologic deficits.
-
Nevoid basal cell carcinoma syndrome - In addition to BCC, this autosomal dominant disorder can result in the early formation of multiple odontogenic keratocysts, palmoplantar pitting, intracranial calcification, and rib anomalies. Various tumors such as medulloblastomas, meningioma, fetal rhabdomyoma, and ameloblastoma also can occur. Pathogenic variants in the PTCH1 or SUFU gene are causative. [37]
-
Rombo syndrome - This autosomal dominant condition is distinguished by BCC and atrophoderma vermiculatum, trichoepitheliomas, hypotrichosis milia, and peripheral vasodilation with cyanosis. [38]
Epidemiology
The American Cancer Society (ACS) reports skin cancer as being the most common cancer in the United States, with basal cell carcinoma (BCC) constituting the majority of cases. The ACS cites an estimate that about 5.4 million basal and squamous cell skin cancers are diagnosed each year in about 3.3 million persons in the US, with about 80% of those being BCCs. Although the number of these skin cancers has been increasing for years, death from them remains uncommon: non-melanoma skin cancers are estimated to cause about 2000-8000 deaths annually (mostly from squamous cell skin cancers), and that number has been decreasing in recent years. [39]
The estimated lifetime risk for BCC in the White population is 33-39% for men and 23-28% for women. BCC incidence doubles every 25 years.
In the US states near the equator, such as Hawaii, BCC incidence is approaching three-fold that of states in the Midwest, such as Minnesota. BCC incidence also varies globally. The highest rates of skin cancer occur in South Africa and Australia, areas that receive high amounts of UV radiation. [40] Australia has a trend toward increasing BCC incidence, while Finland has a low reported incidence that is approximately one quarter that in Minnesota; BCC incidence in Finland also appears to be increasing, however, especially among young women.
BCC is the least likely cancer to metastasize. BCC differs from squamous cell carcinoma, which accounts for 16% of skin cancers and is more life-threatening.
Race
Although BCC is observed in people of all races and skin types, dark-skinned individuals are rarely affected; BCC is most often found in light-skinned individuals (type 1 or type 2 skin). Those with type 1 skin are very fair and have red or blond hair and freckles; these individuals always burn and never tan. Those with type 2 skin are fair and burn easily while tanning minimally. Whites of Celtic ancestry have the highest risk for BCC. Incidence is low in persons with type 5 and 6 skin. [41]
Sex
Historically, men are affected twice as often as women. The higher incidence in men is probably due to increased recreational and occupational exposure to the sun, although these differences are becoming less significant with changes in lifestyle. The current male-to-female ratio is approximately 2.1:1.
For tumors involving the periocular skin, Cook et al reported the incidence of BCC to be equal in men and women. [42] In addition, this investigative team found that the age-adjusted incidence rates for all malignant tumors of the eyelid in men and women, respectively, were 19.6 cases and 13.3 cases per 100,000 population per year. The age-adjusted incidence rates for BCC of the eyelid for men and women, respectively, were 16.9 and 12.4 cases per 100,000 population per year.
Age
The likelihood of developing BCC increases with age. Data indicate that BCC incidence is far higher (more than 100-fold) in persons aged 55-70 years than in those aged 20 years and younger. Patients 50-80 years of age are affected most often. The median age at diagnosis is 67 years and the mean age is 64 years.
Nevertheless, BCC can develop in teenagers and appears frequently in fair-skinned patients aged 30-50 years. Approximately 5% to 15% of cases of BCC occur in patients aged 20-40 years. Aggressive-growth types of BCC are more frequently noted in patients younger than 35 years than in older individuals.
Zhang et al reported an inverse association between body mass index (BMI) and onset of BCC before age 40 years. The multivariate odds ratio for early-onset BCC in obese versus normal individuals was 0.43 for adult BMI and 0.54 for BMI at age 18. [43]
Prognosis
The prognosis for patients with BCC is excellent, with a 100% survival rate for cases that have not spread to other sites. Nevertheless, if BCC is allowed to progress, it can result in significant morbidity, and cosmetic disfigurement is not uncommon.
Typically, basal cell tumors enlarge slowly, relentlessly and tend to be locally destructive. Periorbital tumors can invade the orbit, leading to blindness, if diagnosis and treatment are delayed. BCC arising in the medial canthus tends to be deep and invasive and more difficult to manage; this type of BCC can result in perineural extension and loss of nerve function.
Although BCC is a malignant neoplasm, it rarely metastasizes. The incidence of metastatic BCC is estimated to be less than 0.1%. The most common sites of metastasis are the lymph nodes, lungs, [44] and bones. [45]
Although treatment is curative in more than 95% of cases, BCC may recur, especially in the first year, or develop in new sites. Therefore, regular skin screenings are recommended. [46]
Recurrence
The 5-year recurrence rate is about 5%, but it depends on the histologic subtype and type of treatment; the recurrence rate is less than 1% for primary (previously untreated) BCCs treated with Mohs micrographic surgery. Most reports show that the distance to the closest resection margin is an important predictor of recurrence. [47]
The following is a list of treatments and their 5-year recurrence rates for primary (previously untreated) BCCs:
-
Surgical excision - 10.1%
-
Radiation therapy - 8.7%
-
Curettage and electrodesiccation - 7.7%
-
Cryotherapy - 7.5%
-
All non-Mohs modalities - 8.7%
-
Mohs micrographic surgery - 1%
These rates are probably affected by the fact that clinicians use cryotherapy, curettage, and desiccation mostly on smaller and better-demarcated lesions.
Pieh et al reported a recurrence rate of 5.36% after the first excision of the tumor; the rate increased to 14.7% after the second operation and reached 50% after the third and fourth operations. [48] The highest recurrence, approximately 60%, was seen with lesions arising from the medial canthus.
Recurrences usually occur 4-12 months after initial treatment. One meta-analysis found that the 3-year cumulative risk of devloping a second BCC after an index BCC is about 44%, which is a 10-fold increase over that of the general population. [49, 50] Tumors on the nose or T-zone of the face have a higher incidence of recurrence. Recurrence is most common on the nose and nasolabial fold, but this observation may be secondary to lack of adequate margins obtained in these areas. Infiltrative, micronodular, and multifocal types are more likely than nodular types to recur.
A recurrence of BCC should be suspected when one of the following conditions occurs:
-
Nonhealing ulceration
-
Tissue destruction
-
Scar that becomes red, scaled, or crusted or enlarges with large adjacent telangiectasia
-
Scar that slowly enlarges over time (months)
-
Development of papule/nodule within a scar
Histologic types of BCC at higher risk for recurrence include morpheaform (sclerotic), micronodular, infiltrative, and superficial (multicentric). Higher recurrence rates are also seen with the following:
-
Recurrent tumors that have been treated previously
-
Large tumors (> 2 cm)
-
Deeply infiltrating tumors
Patient Education
Adequate patient education is essential in the prevention of recurrence and spread of basal cell carcinoma. Patients should avoid possible potentiating factors (eg, sun exposure, ionizing radiation, arsenic ingestion, tanning beds). The regular use of sun-protecting clothing (eg, wide-brimmed hat, long-sleeved shirts, sunglasses with ultraviolet [UV] protection) is recommended when outdoors.
Instruct patients to avoid sun exposure particularly during the middle of the day (ie, 11 am to 3 pm), which is the most dangerous time. Also, the sun's rays are especially intense in sunny climates and at high altitudes, and UV radiation can also pass through clouds and water. Patients should be instructed to be careful on the beach and in the snow because sand, water, and snow reflect sunlight and increase the amount of received UV radiation. [51]
During the initial consultation, the patient should be counseled regarding the extent of resection, type of reconstructive procedure, and attendant morbidity. High importance should be attached to adequately preparing the patient regarding the cosmetic and functional result of treatment. During posttreatment follow-up, the patient should be counseled regarding sunlight exposure and the risk of developing additional primary skin tumors.
Sunscreen
Regular application and reapplication of sunscreen is recommended prior to sun exposure. People who use sunscreens have a 40% reduction in skin cancer incidence versus nonusers.
Note that the sun protection factor (SPF) ratings of sunscreens correspond to their ability to protect the skin from harmful UVB rays. The Centers for Disease Control and Prevention (CDC) recommends use of a sunscreen with an SPF rating of at least 15, [52] while the American Academy of Dermatology (AAD) advises use of a sunscreen with an SPF rating of at least 30. [53] Both organizations recommend use of a broad spectrum sunscreen, which provides protection against both UVA and UVB radiation.
Emphasize also that sunscreens must be applied generously, 20-30 minutes before going outside, and reapplied about every 2 hours, more often if swimming or sweating; the AAD recommends use of water-resistant sunscreen, which maintains its SPF for 40 minutes of immersion (or, in the case of "very water resistant" products, for 80 minutes). For lip protection, a lip balm with an SPF of 15 or higher should be applied.
Instruct parents to protect their children's skin with sunscreen or protective clothing to reduce the risk of BCC later in life. It has been estimated that intensive sun protection before age 18 years can reduce nonmelanoma skin cancer by 78%.
Advise parents not to expose children younger than 12 months to direct sunlight and to cover up children aged 12-24 months with a hat, shirt, and a small amount of sunscreen on the remaining exposed areas. Similarly, for children older than 2 years, instruct parents to consider using sunscreens, covering the child's skin with clothing, and, when possible, restricting the child to shaded areas.
Self-examination for skin changes
Educate patients on how to recognize any unexplained changes in their skin, especially changes that last for more than 3-4 weeks. Also, educate patients on how to examine their own and their partner's skin. The knowledge of mole distribution on the skin is helpful.
Tell the patient to first look at the front and back of his or her body in a full-length mirror, using a hand mirror. The patient also should use the hand mirror to look at the back of the neck and scalp, the back, and the breeches. The patient then should turn and look at each side of the body with the arms raised. Next, the patient should bend the elbows and look carefully at the forearms, the back of the upper arms, and the palms. Instruct the patient to sit down and check the backs of the legs and feet, including the spaces between the toes and bottoms of the feet.
The American Cancer Society also offers instructions on how to conduct a skin self-exam.
For additional patient education information, see Skin Cancer and Basal Cell Carcinoma.
-
A pink, scaly lesion on the skin. Superficial basal cell carcinoma (BCC) is often misdiagnosed as eczematous dermatitis or guttate psoriasis and is often difficult to distinguish clinically from Bowen disease (squamous cell carcinoma in situ). Features that suggest the diagnosis of superficial BCC are the absence of significant white, adherent scale and a history of the lesion remaining unchanged for several months or years. Treatment options for this tumor include electrodesiccation and curettage, surgical excision, cryosurgery, 5-fluorouracil, 5% imiquimod cream, and superficial radiotherapy. Electrodesiccation and curettage is the modality most commonly used, with a cure rate of approximately 95%.
-
Basal cell carcinoma.
-
A 68-year-old patient presenting with an advanced basal cell carcinoma (BCC) of the right periorbital region, frontal view. Courtesy of M Abraham Kuriakose, DDS, MD.
-
Lateral view of face showing extent of tumor. Courtesy of M Abraham Kuriakose, DDS, MD.
-
Basal cell carcinoma of the right lower lid.
-
Biopsy-proven basal cell carcinoma of the upper lid margin. Note the loss of cilia (madarosis) in the area of the tumor.
-
Medial canthal/lower lid basal cell. Note the pearly nodular surface with characteristic telangiectatic vessels. Proximity to the lacrimal system will impact its treatment and reconstruction.
-
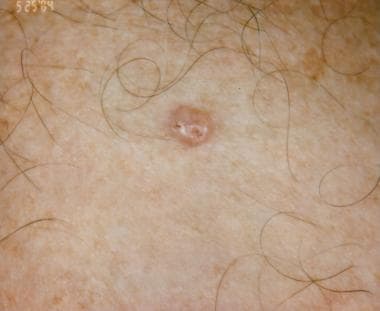
Nodular basal cell carcinoma.
-
Nodular basal cell carcinoma appearing as a waxy, translucent papule with central depression and a few small erosions.
-
Scale, erythema, and a threadlike raised border are present in this superficial basal cell carcinoma on the trunk.
-
Large, superficial basal cell carcinoma.
-
Basal cell carcinoma. Courtesy of Hon Pak, MD.
-
Pigmented basal cell carcinoma.
-
Pigmented basal cell carcinoma.
-
Pigmented basal cell carcinoma has features of nodular basal cell carcinoma with the addition of dark pigmentation from melanin deposition. The pigmentation often has the appearance of dark droplets in the lesion, as shown here.
-
This infiltrating basal cell cancer has ill-defined borders and telangiectases.
-
This translucent pink papule has telangiectases and a crusted erosion, characteristic of nodular basal cell carcinoma.
-
Large, scarlike morpheaform basal cell cancer.
-
Nodular basal cell carcinoma. Nodular aggregates of basalioma cells are present in the dermis and exhibit peripheral palisading and retraction artifact. Melanin is also present within the tumor and in the surrounding stroma, as seen in pigmented basal cell carcinoma.
-
Histologic pattern of a well-differentiated basal cell carcinoma (original magnification X140). Courtesy of Prof Pantaleo Bufo, University of Foggia, Italy.
-
Histologic pattern of a well-differentiated basal cell carcinoma (original magnification X250). Courtesy of Prof Pantaleo Bufo, University of Foggia, Italy.
-
Micronodular basal cell carcinoma often has an absence of retraction artifact. The characteristic histology is small size and uniformity of the tumor nodules. Courtesy of Shang I Brian Jiang, MD.
-
Infiltrative basal cell carcinoma. Tumor cells are arranged in narrow strands, and mucin-rich stroma is often present. Courtesy of Shang I Brian Jiang, MD.
-
Keratotic basal cell carcinoma. Rare type characterized by keratocysts. Courtesy of Shang I Brian Jiang, MD.
-
Basosquamous basal cell carcinoma. Foci of neoplastic cells with squamous differentiation are present. Courtesy of Shang I Brian Jiang, MD.
-
Histology of superficial basal cell carcinoma. Nests of basaloid cells are seen budding from the undersurface of the epidermis. Courtesy of Michael L Ramsey, MD.