Background
Bacterial pneumonia with associated pleural empyema is the most common cause of pleural effusion found in the pediatric population. Parapneumonic effusions are predominately exudative and occur in as many as 50-70% of patients admitted with a complicated pneumonia.
See the image below.
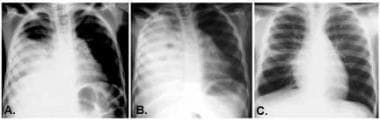 Most parapneumonic effusions treated with the appropriate antimicrobials of sufficient duration resolve without the development of complications or sequelae. The series of radiographs represent a patient treated with thoracentesis alone. Figure A illustrates the patient at presentation. Note the amount of fluid present. In Figure B, the radiograph demonstrates progression of the pleural fluid accumulation with further airspace disease and scoliosis noted. Despite the radiographic evidence of disease progression, the patient was clinically improving. Figure C illustrates the radiograph at follow-up, 6 months following completion of therapy. Resolution of the parapneumonic effusion with no evidence of pleural thickening or fibrosis occurred.
Most parapneumonic effusions treated with the appropriate antimicrobials of sufficient duration resolve without the development of complications or sequelae. The series of radiographs represent a patient treated with thoracentesis alone. Figure A illustrates the patient at presentation. Note the amount of fluid present. In Figure B, the radiograph demonstrates progression of the pleural fluid accumulation with further airspace disease and scoliosis noted. Despite the radiographic evidence of disease progression, the patient was clinically improving. Figure C illustrates the radiograph at follow-up, 6 months following completion of therapy. Resolution of the parapneumonic effusion with no evidence of pleural thickening or fibrosis occurred.
The pulmonary infections of these patients extend into the pleural space and often require more extensive therapy, with associated increased morbidity and extended hospital stay. Involvement of the pleural space with pulmonary infections has been recognized since ancient times. Aristotle identified the increased morbidity and mortality associated with empyema and described drainage of empyema fluid with incision.
The practice of surgical drainage as part of therapy for empyema has continued into the era of modern medicine. In his 1901 text, The Principles and Practice of Medicine, Sir William Osler, MD, stated that empyema should be treated as an ordinary abscess, "with incision and drainage." [1] Of note, Osler underwent a rib resection for his own postpneumonic empyema, from which he ultimately expired.
Complicated parapneumonic effusions are appearing more frequently by most accounts, with reported increases in incidence rates in both Europe and the United States. In England, the rate of admission with a diagnosis of empyema has increased, most notably in children aged 1-4 years. In addition, the identification of Streptococcus pneumoniae as the primary pathogen has also been reported, both in the United States and abroad. [2] Whereas the overall rate of parapneumonic effusions may have stabilized, the rate of bacterial resistance, specifically methicillin-resistant Staphylococcus aureus, has predominated. [3]
Pathophysiology
The definition of a parapneumonic effusion is a pleural effusion associated with either bacterial pneumonia or lung abscess or, rarely, external introduction of organisms associated with chest wall trauma. The development of parapneumonic effusions is gradual, with the pleural fluid collection most commonly divided into 3 stages. The progression of pleural fluid collection evolves from stage 1-3.
In stage 1, the exudative stage, the pleural inflammation from a contiguous infection results in increased permeability and a small fluid collection. At this stage, the effusion is thin and amenable to thoracentesis alone, contains neutrophils, has normal pH and glucose levels, and is often sterile. Stage 2, the fibrinopurulent stage, is characterized by invasion of the organism into the pleural space, progressive inflammation, and significant polymorphonuclear (PMN) leukocyte invasion. The increase in fibrin deposition also results in partitions or loculations within the pleural space. Inflammation is characterized by progressive decreases in the pleural fluid glucose and pH levels and increased protein and lactate dehydrogenase (LDH) levels. The last stage, stage 3, is the organizing stage, in which a pleural peel is created by the resorption of fluid and is associated with fibroblast proliferation that can result in parenchymal entrapment. [4]
Empyema is defined by the presence of intrapleural pus and, for definition purposes, represents an advanced parapneumonic effusion. Complicated parapneumonic effusions (CPE) refer to those fluid collections that require thoracentesis, tube thoracostomy, or surgery for their resolution.
The accumulation of pleural effusions can rapidly occur in the presence of infection. The pleural surface is a mesothelial membrane that covers the lungs and chest wall. The resultant pleural space is a potential space, containing only small volumes of transudative fluid, with a protein content of less than 1.5 g/dL. This fluid is normally composed of lymphocytes, macrophages, and mesothelial cells, with an absence of neutrophils. Interaction of the mesothelium with the invading bacteria, PMNs, and resultant inflammatory mediators can increase pleural permeability. Further PMN recruitment ensues, which results in the increased production of neutrophil chemotactic mediators, ultimately leading to significant pleocytosis.
Activation of the coagulation cascade is common with pleural empyema. The pleural inflammatory response favors increased procoagulant activity, as well as depressed fibrinolytic activity, which favors fibrin deposition. Loculations result with these fibrin strands covered rapidly by a meshwork of fibroblasts that both proliferate and deposit basement membrane proteins onto the surface of the pleura. These proteins obscure the separation of the visceral and parietal pleura and lead to the formation of the pleural peel.
Following initiation of appropriate therapy, the inflammatory cellular and cytokine production recedes, and the PMN predominance of the parapneumonic effusion decreases. An influx of macrophages assists in the clearance of PMNs, with resolution of the inflammatory process. Migration of pleural mesothelial cells into areas of denuded mesothelium results in the reepithelialization of the pleura and recovery of normal function; however, following exuberant pleural inflammation, the potential for pleural fibrosis and restrictive lung disease is enhanced. The mechanisms that lead to either the development of pleural fibrosis or pleural repair with normal recovery are not well understood and need further investigation and characterization.
Etiology
Increased pleural permeability associated with pneumonia or lung abscesses, as well as contiguous infections of the esophagus, mediastinum, or subdiaphragmatic region, may extend to involve the pleura. Similarly, retropharyngeal, retroperitoneal, or paravertebral processes may extend to adjacent structures and involve the pleura, as well.
Host factors that contribute to alterations in pleural permeability, such as noninfectious inflammatory diseases, infection, trauma, or malignancy, may allow accumulation of fluid in the pleural space, which becomes secondarily infected.
The bacteriology of the pleural space varies with patient age. In the pediatric population, the most common implicated organisms are S pneumoniae, S aureus, and group A streptococci. H influenzae is rarely observed since the advent of the H influenzae B vaccine.
Because of the use of oral antibiotics before the recognition of the parapneumonic effusion, most specimens cultured are sterile; thus, the relative incidences of the aforementioned organisms are not known.
Anaerobic infections secondary to aspiration and fungal or mycobacterial infections in immunosuppressed patients are also reported.
Mycoplasma pneumoniae, viruses, and atypical pneumonias can also present with exudative pleural effusions, although mononuclear cells primarily characterize them.
A retrospective study of pediatric empyema conducted in Australia found that S pneumoniae was the most common pathogen (75/195, 39%), with serotype 3 the most frequently detected (27/75, 27%). S aureus occurred more often in infants and children of indigenous background, compared with S pneumoniae. [5]
A subsequent study in Australia showed that the incidence and severity of empyema among pediatric patients have increased in the period following the COVID-19 pandemic. During this period, the rate of Streptococcus pyogenes infection has also risen. [6]
A study investigated the risk factors of empyema after acute viral infection and the association between empyema and the use of nonsteroidal anti-inflammatory drugs (NSAIDs). The study found that NSAID use during acute viral infection is associated with an increased risk of empyema in children. [7]
Epidemiology
United States statistics
Parapneumonic effusions are predominately exudative and occur in as many as 50-70% of patients admitted with a complicated pneumonia. These patients have extension of their pulmonary infection into the pleural space and require more extensive therapy, with associated increased morbidity and extended hospital stay. Incidence rates remain at approximately 10 cases per 10,000. [3]
International statistics
With an increased incidence of pneumonia and tuberculosis worldwide, the frequency of complicated parapneumonic effusions (CPE) is likely to be even higher. Literature emerging from Asia suggests empyema is a significant concern, although these retrospective reviews cannot accurately describe the incidence, and variable rates of identification suggest more standardized practices are needed. [8]
Age-related demographics
The bacteriology of the infection varies with patient age. In the pediatric population, the most commonly implicated organisms are S pneumoniae, S aureus, and group A streptococci. The latter may be observed as a complication of an infectious skin disorder, such as varicella, impetigo, or infectious eczema. Haemophilus influenzae is rarely encountered since the advent of the H influenzae B vaccine. Methicillin-resistant S aureus is a concern in the older age group.
Prognosis
The prognosis for most patients with parapneumonic effusions is quite good. Extended antibiotics may be needed in some patients with CPE. Despite the variability in presentation, most patients recover without sequelae.
Numerous studies have demonstrated resolution of the radiographic abnormalities by 3-6 months following therapy, with few to no symptoms reported at follow-up examination.
Pulmonary function testing performed following hospitalization has not shown marked abnormalities, regardless of clinical course. The only abnormality observed may be slight expiratory flow limitation. Mild obstructive abnormalities were the only findings observed in patients evaluated 12 years (±5) following recovery from empyema.
Some increased bronchial reactivity has been reported at later follow-up examinations; however, lung function and exercise response return to normal for most patients.
Early recognition of pneumonia with parapneumonic effusion, effective intervention to identify the causative organism, and initiation of definitive therapy reduce morbidity and complications associated with this process.
Morbidity/mortality
Early recognition of pneumonia with parapneumonic effusion, effective intervention to identify the infecting organism, and initiation of definitive therapy reduce the morbidity and complications associated with this process.
Complications
Fibrothorax, a complication reported in the adult literature, is rarely observed in pediatric patients.
-
Radiographic imaging of a parapneumonic effusion may be useful in assessing the stage of the effusion and the type of drainage needed. In Figure A, the left heart border is obscured, and more than 50% of the left hemithorax is filled with an effusion, as evidenced by a fluid meniscus. In Figure B, the effusion is demonstrated to be fluid because it layers out on a decubitus film. In Figure C, the lateral radiograph again demonstrates the fluid meniscus and filling of the posterior sulcus. These findings suggest tube thoracostomy placement may be sufficient to drain this pleural process.
-
Most parapneumonic effusions treated with the appropriate antimicrobials of sufficient duration resolve without the development of complications or sequelae. The series of radiographs represent a patient treated with thoracentesis alone. Figure A illustrates the patient at presentation. Note the amount of fluid present. In Figure B, the radiograph demonstrates progression of the pleural fluid accumulation with further airspace disease and scoliosis noted. Despite the radiographic evidence of disease progression, the patient was clinically improving. Figure C illustrates the radiograph at follow-up, 6 months following completion of therapy. Resolution of the parapneumonic effusion with no evidence of pleural thickening or fibrosis occurred.


