Definition
Pleomorphic adenoma is a benign salivary gland tumor that exhibits wide cytomorphologic and architectural diversity. The tumor has the following three components: the epithelial cell, myoepithelial cell, and stromal/mesenchymal. Identification of these three components — which may vary quantitatively and qualitatively from one tumor to another — is essential for the recognition of pleomorphic adenoma. Historically, this tumor was referred to as a benign mixed tumor. See Benign Tumors of Major Salivary Glands Clinical Presentation for more information, including tumor classification, presentation, and treatment.
The epithelial component of pleomorphic adenoma forms the inner layer of cysts and tubules, while the myoepithelial cells constitute the outer layer and are scattered within the myxoid stroma. The myoepithelial cells display remarkable morphological diversity, appearing as plasmacytoid, spindled, epithelioid, clear, or stellate-shaped cells. The stromal component is typically myxoid, chondroid, or myxochondroid but can also be hyalinized or fibrotic. [1, 2]
The tumor exhibits a bosselated outer surface, frequently with tongue-like protrusions called pseudopodia that extend beyond the main tumor mass. These pseudopodia represent a significant risk factor for local recurrence following surgical excision. [3]
Epidemiology
Pleomorphic adenoma is the most common salivary gland tumor in both children and adults, accounting for 45-75% of all salivary gland neoplasms. [4, 5] The annual incidence is approximately 2-3.5 cases per 100,000 population. [6] Although these tumors occur across all age groups, they are most common in the third to sixth decades, with an average age at presentation between 43 and 46 years. [7] Pleomorphic adenomas are seen slightly more often in females than males. [8]
Etiology
Although the exact etiology of pleomorphic adenoma is unknown, the incidence has been found to increase 15 to 20 years after exposure to radiation. [4] Genetic predisposition may play a role along with other factors such as cigarette smoke and exposure to toxins. Research suggests both somatic mutation and autosomal dominant inheritance patterns in the causation of pleomorphic adenoma. [2]
Interestingly, the simian virus 40 (SV40), a highly oncogenic tumor virus, has been implicated in the development of pleomorphic adenomas. SV40 tag sequences have been frequently identified by polymerase chain reaction in pleomorphic adenomas but not in normal salivary gland tissue, suggesting this virus may contribute to tumor development or progression. [2]
If left untreated, approximately 6.2% of pleomorphic adenomas may undergo malignant transformation to carcinoma ex pleomorphic adenoma (CXPA). Risk factors for malignant transformation include multiple recurrences, male sex, advanced age, previous radiation therapy, and larger tumor size. [9]
Location
Pleomorphic adenomas occur most often in the parotid gland, with a predilection for the tail of the superficial lobe (70-80% of cases). [10] The tumor is less commonly seen in the submandibular gland (10%), and it is seldom encountered in the sublingual gland (1%). [11, 12]
Pleomorphic adenomas of the minor salivary glands comprise only 5-10% of cases, with the palate (particularly the junction of the soft and hard palate) and the lip being the most common sites. Other sites of minor salivary gland involvement may include the nose, the paranasal sinuses, and the larynx.
Rare or unusual sites of occurrence may be attributable to ectopic salivary gland tissue (e.g., in the mandible, neck lymph nodes, or axilla). A case has even been reported in which a pleomorphic adenoma presented as a midline nodule in the isthmus of the thyroid. [13]
Multiple tumors are unusual (1:40,000), but metachronous and synchronous tumors do occur. [14, 15, 16, 17] Synchronous occurrence has also been reported of pleomorphic adenoma and Warthin's tumor (the second most common benign salivary gland tumor). [18, 19, 20, 21]
Clinical Features and Imaging
Pleomorphic adenomas usually arise as a slow-growing, painless mass, which may be present for many years. Clinically, these tumors manifest as well-delineated, mobile nodules covered with normal mucous membrane, though ulcerations can occasionally be observed. [2]
Signs and symptoms depend on the tumor size and location. [12] Facial nerve weakness and/or rapid growth should raise concern for malignant transformation. Pleomorphic adenomas in the deep lobe of the parotid gland may present as an oral retrotonsillar mass or parapharyngeal space tumor.
Patients with minor salivary gland tumors may present with a variety of symptoms depending on the site of the tumor. These symptoms may include dysphagia, dyspnea, hoarseness, difficulty chewing, and epistaxis.
On ultrasound, pleomorphic adenomas typically appear as hypoechoic lesions with well-defined, lobulated borders, with or without posterior acoustic enhancement. [2]
On computed tomography, these tumors present as globular masses with smoothly marginated or lobulated homogeneous soft tissue density. Larger tumors may exhibit necrosis. Few calcification foci may be seen. Smaller tumors show early homogeneous significant enhancement, while larger tumors demonstrate less pronounced and delayed enhancement. [2]
On magnetic resonance imaging, smaller masses appear well-circumscribed and homogeneous, while larger tumors appear heterogeneous. [2]
Recurrent pleomorphic adenomas characteristically present as multiple nodules of variable size often appearing as solitary or multiple subcutaneous T2 hyperintense lesions in the operative bed, subcutaneous fat, and/or spaces adjacent to the parotid. [22]
For diagnostic confirmation, fine needle aspiration can identify tumor malignancy with approximately 90% sensitivity, while core needle biopsy, though more invasive, provides a more precise determination of the histological type with about 97% diagnostic accuracy. [2]
Gross Findings
On gross examination, pleomorphic adenomas typically appear a solitary, firm, mobile, and well-circumscribed mass that rarely exceeds 6 cm in its greatest dimensions. [1] The color of these tumors varies from white-tan to blue-gray, depending on the presence or absence of cartilaginous features. Their size averages between 2 cm and 6 cm, though pleomorphic adenomas may range from a few millimeters to quite large. [23, 24] Degenerative and cystic changes may be encountered on sectioning. It is not unusual to observe evidence of focal or more extensive infarction, especially in cases with prior fine needle aspiration. Recurrent tumors characteristically present as multiple nodules of variable size.
A thick fibrous capsule is present in approximately 40% of excised cases, and about 20% may have satellite nodules within their capsule, which may contribute to recurrence if simple enucleation is performed. [2]
Microscopic Findings
Despite their highly variable appearance, pleomorphic adenomas can be fundamentally regarded as a "biphasic" tumor as their ducts and tubules exhibit two cell layers: an inner or luminal ductal cell layer and an outer or abluminal myoepithelial cell layer. The varying proportion of these cell types as well as the stromal components accounts for the morphologic diversity of these tumors.
Pleomorphic adenomas with an approximately equal proportion of cellular and stromal elements are referred to as classic or mixed-type tumors. When the cellular elements predominate, the result is an epithelial cell-rich or myoepithelial cell-rich (i.e., cellular) pleomorphic adenoma. When there is a predominance of stroma, the result is a stroma-rich pleomorphic adenoma (see the image below). The variations in cellular to stromal ratios do not appear to impart any clinical difference in tumor behavior. [4] In fact, in many cases, different regions of the same tumor may display a striking diversity in morphology and cellularity.
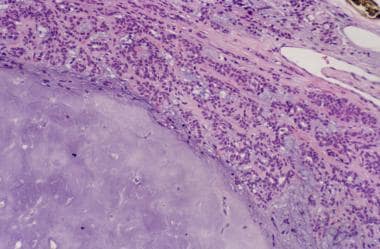 Pathology of pleomorphic adenoma. Variable cellularity in pleomorphic adenoma. The right upper section of the image shows a cellular region with dense epithelial and myoepithelial cells. In the left lower section of the image, the dominant morphology is that of chondroid stroma, with only a few interspersed myoepithelial cells.
Pathology of pleomorphic adenoma. Variable cellularity in pleomorphic adenoma. The right upper section of the image shows a cellular region with dense epithelial and myoepithelial cells. In the left lower section of the image, the dominant morphology is that of chondroid stroma, with only a few interspersed myoepithelial cells.
The cellular component consists of the epithelial and myoepithelial cells with an array of architectural growth patterns, including trabecular, tubular, solid, cystic, and papillary. Both cell types may show a range of phenotypes. The epithelial cells are often cuboidal with small tubule formation. The myoepithelial cells surround the epithelial cells and disperse or "melt" into the surrounding stroma, which may also contain so-called modified myoepithelial cells (see the following image).
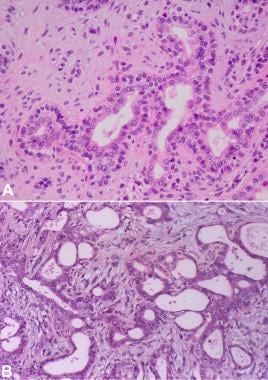 Pathology of pleomorphic adenoma. Cells in pleomorphic adenoma. (A) Tubule/duct formations in pleomorphic adenoma. Well-defined inner epithelial lining, consisting of cuboidal cells with eosinophilic cytoplasm, which can focally be clear, is surrounded by one or more layers of myoepithelial cells with mostly clear cytoplasm. The stroma in between contains dispersed spindled and epithelioid myoepithelial cells. (B) A similar tubular arrangement is seen, but the myoepithelial cells are not as well defined. The stroma in between shows dispersed spindled myoepithelial cells. In both images A and B, note the bland nature of the cells and the absence of atypia.
Pathology of pleomorphic adenoma. Cells in pleomorphic adenoma. (A) Tubule/duct formations in pleomorphic adenoma. Well-defined inner epithelial lining, consisting of cuboidal cells with eosinophilic cytoplasm, which can focally be clear, is surrounded by one or more layers of myoepithelial cells with mostly clear cytoplasm. The stroma in between contains dispersed spindled and epithelioid myoepithelial cells. (B) A similar tubular arrangement is seen, but the myoepithelial cells are not as well defined. The stroma in between shows dispersed spindled myoepithelial cells. In both images A and B, note the bland nature of the cells and the absence of atypia.
Myoepithelial cells may have plasmacytoid, epithelioid, spindled, oncocytic, or clear cell morphology. [8, 25, 26, 27] Some studies have shown plasmacytoid and spindled morphologies, respectively, are the most common. [8, 28] The cellular elements are typically cytologically bland, with no significant mitotic activity. Some have noted apocrine changes—particularly within the ductal elements — which may be on a spectrum with intracapsular CXPA. [29] This apocrine differentiation can be highlighted with immunohistochemical studies for gross cystic disease fluid protein 15 (i.e., GCDFP-15) or androgen receptor.
Pleomorphic adenomas occasionally exhibit unusual components, including sebaceous cells, mucous cells, squamous cells, oncocytic cells, serous acini, fibroadipose tissue, and tyrosine crystals (see the image below). [30, 31]
The presence of mucous cells may prompt the examiner to consider entities such as mucoepidermoid carcinoma, especially in limited specimens. [32, 33] These foci have been shown to be negative for MAML2 translocations, however, and the presence of other features (e.g., stroma and myoepithelial cells) should also help in establishing the correct diagnosis. [29]
 Pathology of pleomorphic adenoma. Unusual findings in pleomorphic adenoma. (A) Tyrosine crystals with the characteristic rosette appearance. (B) Large cystic changes in a pleomorphic adenoma that is rich in myoepithelial cells. The arrow indicates an area of squamous cell metaplasia. (C) Foci of mature adipose tissue in the stroma. Note the background of myoepithelial-rich stroma. (D) Areas of mucous cells (arrows) are present next to epithelial/myoepithelial formations.
Pathology of pleomorphic adenoma. Unusual findings in pleomorphic adenoma. (A) Tyrosine crystals with the characteristic rosette appearance. (B) Large cystic changes in a pleomorphic adenoma that is rich in myoepithelial cells. The arrow indicates an area of squamous cell metaplasia. (C) Foci of mature adipose tissue in the stroma. Note the background of myoepithelial-rich stroma. (D) Areas of mucous cells (arrows) are present next to epithelial/myoepithelial formations.
The stromal component is a product of the modified myoepithelial cells and may appear myxoid, chondroid, chondromyxoid, hyalinized, or even osseous (see the image below). [34, 35] Chondroid areas represent true cartilage formation (containing type II collagen and keratin sulfate); this finding tends to be specific for pleomorphic adenoma and aids in the distinction of these masses from other salivary gland tumors. Squamous metaplasia is a well-documented phenomenon reported in approximately 17.9% of cases. Other metaplasias (e.g., mucinous metaplasia in 2.9% and oncocytic changes in 5.9%) and skin adnexal-type differentiation have also been described. [29, 36, 37] Some authors note that a prominent hyalinized stroma may portend an increased propensity for malignant change. [38]
 Pathology of pleomorphic adenoma. Different types of stroma in pleomorphic adenoma. (A) Myxoid stroma. Note the dispersed spindle myoepithelial cells in the stroma, some of which have stellate morphology; (B) Chondroid stroma; (C) Stroma with cartilage formation. (D) Hyalinized stroma.
Pathology of pleomorphic adenoma. Different types of stroma in pleomorphic adenoma. (A) Myxoid stroma. Note the dispersed spindle myoepithelial cells in the stroma, some of which have stellate morphology; (B) Chondroid stroma; (C) Stroma with cartilage formation. (D) Hyalinized stroma.
In the parotid, pleomorphic adenomas are usually surrounded by a fibrous capsule of variable thickness that may be focally discontinuous, especially in more stromal-rich tumors (see the following image). In minor salivary glands, a capsule is characteristically absent. Pleomorphic adenomas may demonstrate microscopic satellite tumor nodules beyond the capsule, pseudopodia, and capsular penetration; these findings are the purported causes of recurrence in cases treated with simple enucleation or cases in which surgical resection was performed with inadequate surgical margins (see the image below). [39, 40, 41]
 Pathology of pleomorphic adenoma. Capsule in pleomorphic adenoma (PA). (A) A PA with a thin fibrous capsule separating the tumor from the surrounding normal parotid gland tissue. (B) A PA with a less well developed/focally absent capsule. Note the small satellite tumor nodule (arrow) protruding outside a major tumor mass.
Pathology of pleomorphic adenoma. Capsule in pleomorphic adenoma (PA). (A) A PA with a thin fibrous capsule separating the tumor from the surrounding normal parotid gland tissue. (B) A PA with a less well developed/focally absent capsule. Note the small satellite tumor nodule (arrow) protruding outside a major tumor mass.
 Pathology of pleomorphic adenoma. Recurrent pleomorphic adenoma (PA) with two microscopically benign–appearing nodules. Multiple nodules are a characteristic pattern in recurrent PAs.
Pathology of pleomorphic adenoma. Recurrent pleomorphic adenoma (PA) with two microscopically benign–appearing nodules. Multiple nodules are a characteristic pattern in recurrent PAs.
Cystic changes are occasionally seen in pleomorphic adenomas. A large cyst may be present, or the tumor may be multicystic. Such changes do not appear to have an impact on the clinical course of the tumor.
Necrosis and increased mitosis are not commonly encountered in pleomorphic adenomas; when they do occur, a thorough search for evidence of malignant change is necessary. Fine needle biopsy or focal infarction may result in similar tumoral changes, and this possibility should also be carefully considered. Other atypical findings, such as increased cellularity, focal atypia, pleomorphism, and capsular extension, should not be interpreted as indicating malignant change in the absence of other corroborating evidence.
Occasionally (i.e., in 1-3% of pleomorphic adenomas), benign tumor fragments are seen within vascular spaces in the tumor or in the immediate peritumoral vasculature. These deposits are thought to result from perioperative manipulation of the tumor during surgical resection and therefore should not be interpreted as a malignant feature. [29, 35]
Immunohistochemistry
In most instances, the diagnosis of pleomorphic adenoma is made through straightforward microscopic identification; however, immunohistochemistry (IHC) may be utilized to delineate the different cell types and components and to aid in differentiating pleomorphic adenoma from other tumor types. [29, 42, 43, 44] The following IHC stains have proven helpful:
-
Low molecular weight cytokeratins, epithelial membrane antigen - Positive in luminal epithelial cells
-
High molecular weight cytokeratins - Positive in abluminal myoepithelial/basal cells
-
Cytokeratin 7 - Shows diffuse and strong immunoreactivity in the lining epithelial cells [45]
-
Carcinoembryogenic antigen - Positive in the luminal border of the ductal component [46]
-
p63, p40 - Positive in abluminal myoepithelial/basal cells; usually concordant labeling [47]
-
Sox10 - Positive in abluminal myoepithelial/basal cells
-
Muscle markers (e.g., calponin, smooth muscle actin, smooth muscle myosin heavy chain) - Positive in myoepithelial cells
-
S100 - Positive in myoepithelial cells
-
Glial fibrillary acidic protein - Positive in myoepithelial cells in myxoid areas
-
Pleomorphic adenoma gene 1 (PLAG1) - Sensitive but not specific for PLAG1 fusion
Molecular/Genetics
The majority of pleomorphic adenomas show cytogenetic aberrations, which may manifest as follows: [5, 27, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57]
-
PLAG1 rearrangements (50-60%) - rearrangement involving 8q12 with various partners (CTNNB1, FGFR1, LIFR, CHCHD7, or TCEA1)
-
HMGA2 rearrangements (20-30%) - rearrangement involving 12q14-15 (high mobility group AT-hook 2 gene, HMGA2) with various partners (NFIB, FHIT, and WIF1)
-
Sporadic, non-clonal changes due to intrachromosomal rearrangements
-
RNA sequencing analyses have identified numerous previously unreported gene fusions involving PLAG1 and HMGA2, with 15 novel fusions documented in a 2025 comprehensive study [58]
-
Cryptic rearrangements - Importantly, PLAG1 and HMGA2 fusions have been detected in tumors with normal karyotypes, demonstrating that these genetic alterations can arise from cryptic intra- or interchromosomal rearrangements that are undetectable by conventional cytogenetic analysis [58]
A study on transcriptomic profiling has revealed two distinct molecular subclusters of pleomorphic adenomas characterized by PLAG1-activation and HMGA2-activation, respectively. These subclusters show differential expression of genes involved in immune system function, cell adhesion, and microenvironment remodeling. [58]
Specific fusion variants may correlate with distinct clinicopathological features. For example, the TGFBR3-PLAG1 fusion has been characterized in myoepithelial carcinomas and CXPAs, with these tumors showing a predilection for the deep lobe of the parotid gland and the potential for high-grade transformation. [59]
As stated above, IHC for PLAG1 is sensitive but not specific for pleomorphic adenomas and CXPAs. Conversely, IHC for HMGA2 appears specific but not sensitive for these tumor types. Both seem to correlate with their respective rearrangements. PLAG1 and HMGA2 fusions are unique to pleomorphic adenomas and CXPAs, though they do not distinguish benign (i.e., pleomorphic adenomas) from malignant (i.e., CXPAs) tumors, and so their diagnostic utility is somewhat limited. They may aid in identifying morphologic mimics or unusual tumor morphologies. For example, HMGA2-WIF1 rearrangements have been described in pleomorphic adenomas with a stroma-poor, "canalicular adenoma-like" appearance. [60] Lastly, molecular findings may be useful in confirming an originating pleomorphic adenoma component in CXPAs, in which the classic pleomorphic adenoma architecture is hyalinized or otherwise indiscernible.
Less commonly described molecular events include mutation and overexpression of HRAS, [27] the mucin 1 gene (MUC1) associated with recurrence and malignant transformation of pleomorphic adenomas (with carcinoma cells overexpressing MUC1), [61, 62] and a trend toward the presence of BRCA1/2 mutations in pleomorphic adenomas and CXPAs. [63]
In the progression from pleomorphic adenoma to CXPA, molecular studies support a multi-step model of carcinogenesis. The malignant transformation involves additional genetic alterations including TP53 mutations, PIK3R1 frameshift mutations, and various allelic imbalances. [37]
A documented case of sequential pleomorphic adenoma to CXPA transformation revealed that the same LIFR/PLAG1 translocation and PIK3R1 mutation were present in both the original pleomorphic adenoma and subsequent malignant recurrences, with additional TP53 mutations appearing only in the malignant lesions. [37]
Studies have also identified upregulation of genes involved in extracellular matrix production, WNT signaling, and epithelial-mesenchymal transition in pleomorphic adenomas compared to normal salivary tissue. [58] Additionally, there appears to be a trend toward the presence of BRCA1/2 mutations in both pleomorphic adenoma and CXPA samples, with significant differences in expression patterns between these entities. [64]
Prognosis and Predictive Factors
For most patients with benign, well-circumscribed pleomorphic adenomas, the prognosis is excellent after surgical resection with low rates of recurrence. However, recurrence can be a problem, and it may be related to an incomplete tumor capsule, extension of tumor nodules beyond the capsule (i.e., pseudopodia), the presence of separate satellite nodules, and intraoperative tumor rupture/disruption. [29, 64, 65] Recurrences usually present in a multinodular fashion. [66, 67] Other factors associated with recurrence include tumors with a high mesenchymal content, female sex, younger patients, and surgical enucleation (vs parotidectomy). [27, 68, 69]
Malignant transformation occurs in approximately 6% of pleomorphic adenomas, which justifies their resection. [27] Multiple recurrences, deep lobe tumors, male sex, and older age have all been associated with an increased risk of malignancy. Sometimes malignant features may be subtle and only realized after thorough sampling and evaluation. [70, 71, 72]
Rarely, these tumors may metastasize. Metastasizing pleomorphic adenomas are histologically indistinguishable from pleomorphic adenomas. Further, some have been shown to harbor the same PLAG1 and HMGA2 molecular alterations as non-metastasizing pleomorphic adenomas. [73] Metastasizing pleomorphic adenomas notoriously occur after multiple recurrences, with the most common sites being the regional lymph nodes, bone, and lung. [27] Possible mechanisms for this behavior include vascular permeation of the tumor (either de novo or as a consequence of surgical manipulation) or an inherent but low-risk potential in a subset of tumors. As such and due to the occasional aggressive course, it is recommended to avoid labeling these tumors as benign. [29, 73]
-
Pathology of pleomorphic adenoma. Capsule in pleomorphic adenoma (PA). (A) A PA with a thin fibrous capsule separating the tumor from the surrounding normal parotid gland tissue. (B) A PA with a less well developed/focally absent capsule. Note the small satellite tumor nodule (arrow) protruding outside a major tumor mass.
-
Pathology of pleomorphic adenoma. Different types of stroma in pleomorphic adenoma. (A) Myxoid stroma. Note the dispersed spindle myoepithelial cells in the stroma, some of which have stellate morphology; (B) Chondroid stroma; (C) Stroma with cartilage formation. (D) Hyalinized stroma.
-
Pathology of pleomorphic adenoma. Cells in pleomorphic adenoma. (A) Tubule/duct formations in pleomorphic adenoma. Well-defined inner epithelial lining, consisting of cuboidal cells with eosinophilic cytoplasm, which can focally be clear, is surrounded by one or more layers of myoepithelial cells with mostly clear cytoplasm. The stroma in between contains dispersed spindled and epithelioid myoepithelial cells. (B) A similar tubular arrangement is seen, but the myoepithelial cells are not as well defined. The stroma in between shows dispersed spindled myoepithelial cells. In both images A and B, note the bland nature of the cells and the absence of atypia.
-
Pathology of pleomorphic adenoma. Variable cellularity in pleomorphic adenoma. The right upper section of the image shows a cellular region with dense epithelial and myoepithelial cells. In the left lower section of the image, the dominant morphology is that of chondroid stroma, with only a few interspersed myoepithelial cells.
-
Pathology of pleomorphic adenoma. Recurrent pleomorphic adenoma (PA) with two microscopically benign–appearing nodules. Multiple nodules are a characteristic pattern in recurrent PAs.
-
Pathology of pleomorphic adenoma. Unusual findings in pleomorphic adenoma. (A) Tyrosine crystals with the characteristic rosette appearance. (B) Large cystic changes in a pleomorphic adenoma that is rich in myoepithelial cells. The arrow indicates an area of squamous cell metaplasia. (C) Foci of mature adipose tissue in the stroma. Note the background of myoepithelial-rich stroma. (D) Areas of mucous cells (arrows) are present next to epithelial/myoepithelial formations.


